Design of an internal salt bridge in zwitterionic SUCNR1-antagonists for improved oral exposure
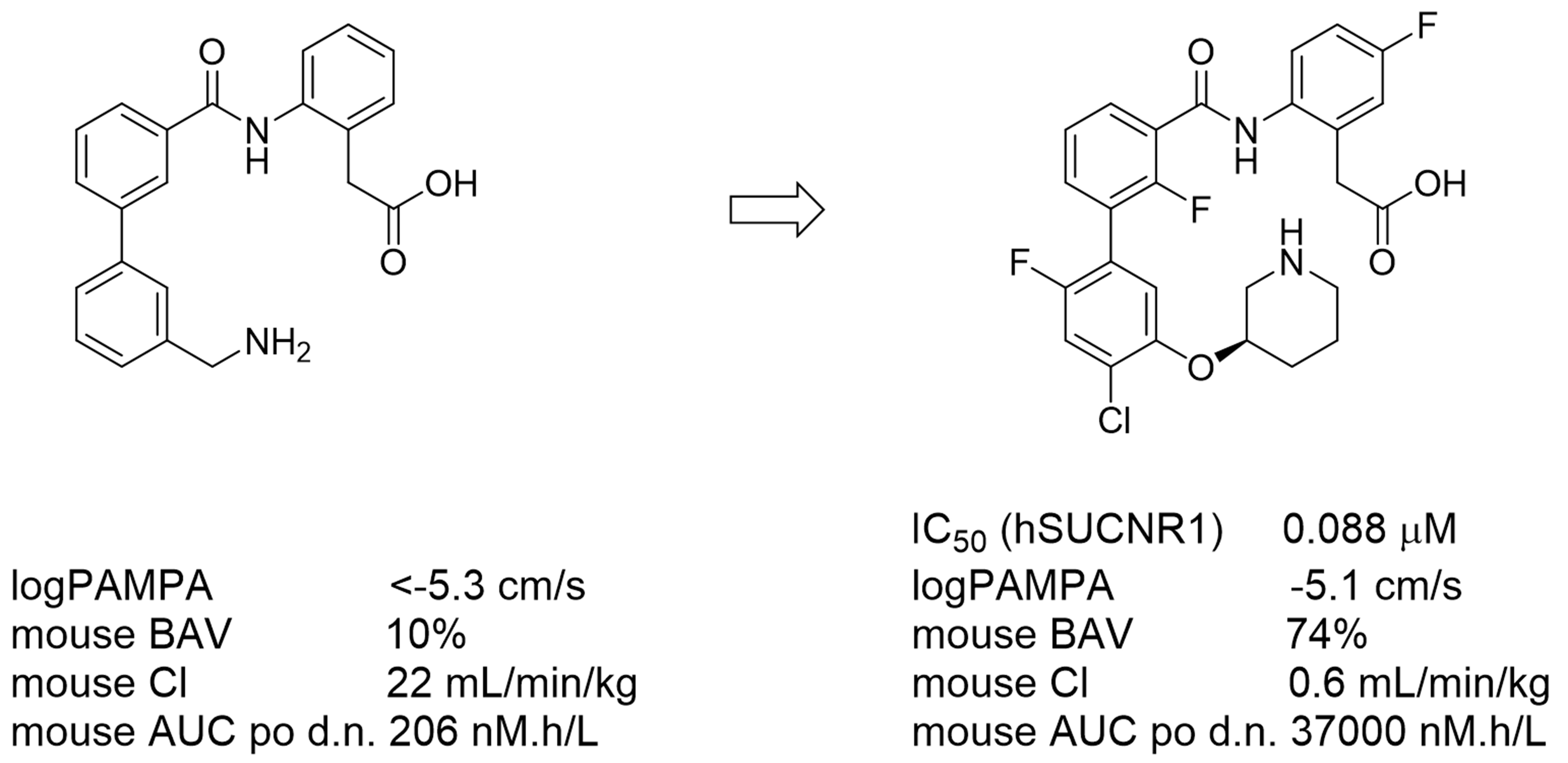
SUCNR1 (succinate receptor 1 or GPR91) is a G-protein coupled receptor sensing succinate, a signaling molecule when released into the extracellular space under certain pathological conditions.1 SUCNR1 is implicated in various pathological conditions such as liver fibrosis, obesity or atherothrombosis and plays also an important role in inflammation.2 Recently, a novel SUCNR1 antagonist scaffold has been discovered by our group by high-throughput screening.3 The hit-to-lead optimization led to zwitterionic compounds which, however, suffered from poor absorption properties. In order to improve the absorption and oral exposure, formation of an internal salt bridge was envisioned for shielding of both opposite charges and as a consequence, the high polarity found in zwitterions with separated charges. Guided by a pair of rather close zwitterionic analogs with very different permeation and absorption properties new, potent, salt bridge-containing SUCNR1 inhibitors were designed. The new analogs demonstrated indeed much higher oral bioavailability and oral exposure. Importantly, such compounds were found to be of a chameleonic nature, i.e. they form the salt bridge allowing them for good permeation in their unbound form while the salt bridge can be disrupted in their bound form which allows for their binding into the receptor with both ions required to be separated.3
[1] Robert Haas, Danilo Cucchi, Joanne Smith, Valentina Pucino, Claire Elizabeth Macdougall, Claudio Mauro, Trends Biochem. Sci. 2016, 41, 460– 471.
[2] Grzegorz Krzak, Cory M. Willis, Jayden A. Smith, Stefano Pluchino, Luca Peruzzotti-Jametti, Trends Immunol., 2021, 42, 45−58.
[3] Juraj Velcicky, Rainer Wilcken, Simona Cotesta, Philipp Janser, Achim Schlapbach, Trixie Wagner, Philippe Piechon, Frederic Villard, Rochdi Bouhelal, Fabian Piller, Stephanie Harlfinger, Rowan Stringer, Dominique Fehlmann, Klemens Kaupmann, Amanda Littlewood-Evans, Matthias Haffke, Nina Gommermann, J. Med. Chem. 2020, 63, 9856−9875.